Gap Analysis Remediation: A Guide to Resourcing & Implementationįrom the very start, it's important to make sure everything is pointed at the same target. Planning a quality system gap analysis? Grab our free guide and learn how to turn your finished report into an actionable project plan: This starter guide offers some helpful guidance to help you orient yourself in the initial planning stage of such a project and how expert third party resources play an important role in ensuring these assessments are effective and efficient. Between planning, coordinating, and conducting the assessment, audits like this have many moving parts, each of which critical to the project's success. 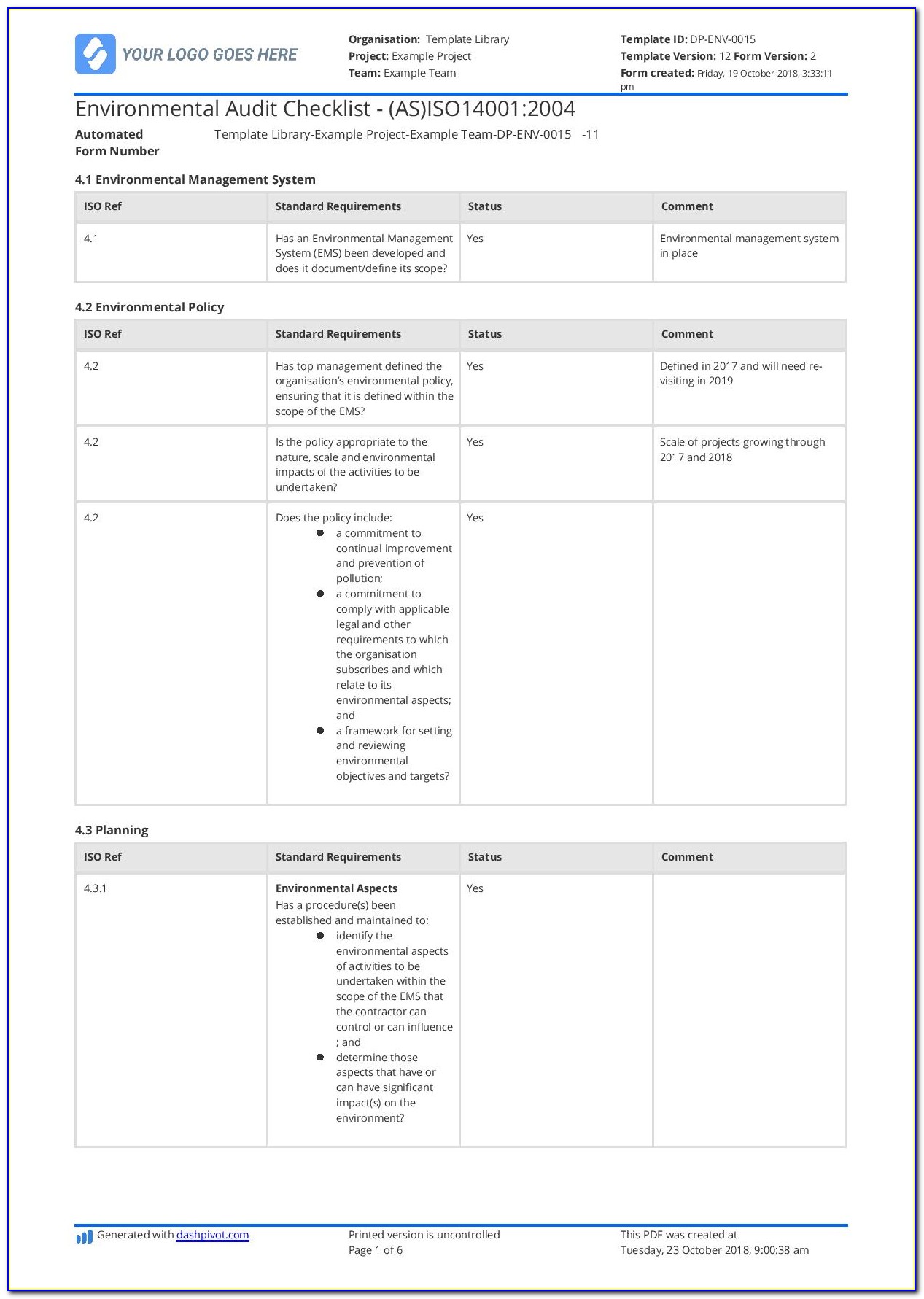
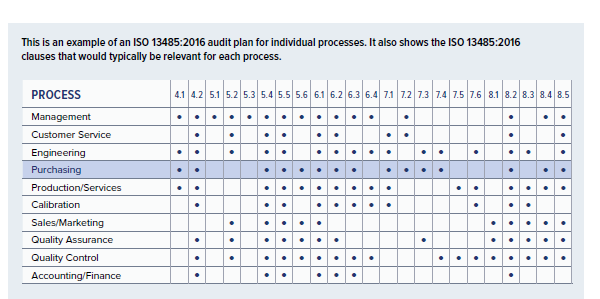
These audit programs can be daunting and complex, making it difficult to know where to start. Medical device manufacturers and supporting organizations pursuing or maintaining an ISO 13485:2016 certification must conduct regular audits to ensure an adequate, effective quality system is established and maintained through a compliant Quality Management Systems (QMS).
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
